Breast augmentation is the most prevalent cosmetic procedure in the United States, with estimates ranging that 1 to 3 million women have some type of breast implant. For years, there has been developing research about the possibility of implants increase the risk of certain cancers, but the evidence was considered too slim to create any action towards addressing the problem.
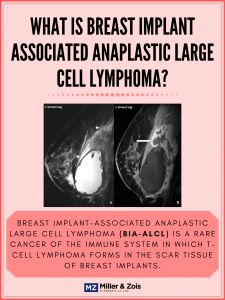 Now, the FDA has finally issued a safety warning about the risks of using breast implants manufactured by the company Allergan. The warning requested Allergan to recall all of their micro-textured implants in the United States, citing new sufficient evidence showing a link between their products and breast implant-associated anaplastic large cell lymphoma (BIA-ALCL). This recall will most likely cause a significant increase in breast implant cancer lawsuits, including class action lawsuits for medical monitoring.
Now, the FDA has finally issued a safety warning about the risks of using breast implants manufactured by the company Allergan. The warning requested Allergan to recall all of their micro-textured implants in the United States, citing new sufficient evidence showing a link between their products and breast implant-associated anaplastic large cell lymphoma (BIA-ALCL). This recall will most likely cause a significant increase in breast implant cancer lawsuits, including class action lawsuits for medical monitoring.
The warning is not without precedent, as there have been similar recalls for Allergan breast implants in Canada, Australia, and France within the past year. However, the FDA was hesitant to issue on their own recall due to “insufficient evidence” that indicated a connection between textured implants and the rare lymphoma cancer. Despite this, hundreds of women around the country have been taking Allergan to court based on claims that their breast implants were the cause of their illness.





